Monosodium arsenate
Sodium dihydrogen arsenate
CAS: 10103-60-3
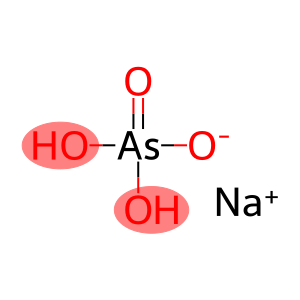
Molecular Formula: AsH2NaO4
Monosodium arsenate - Names and Identifiers
| Name | Sodium dihydrogen arsenate |
| Synonyms | Ai3-01067 Monosodium arsenate Sodium arsenate monobasic Sodium arsenate (nah2aso4) Sodium dihydrogen arsenate Sodium dihydrogen orthoarsenate Arsenic acid (H3aso4), monosodium salt |
| CAS | 10103-60-3 |
| EINECS | 231-547-5 |
| InChI | InChI=1/AsH3O4.Na/c2-1(3,4)5;/h(H3,2,3,4,5);/q;+1/p-1 |
Monosodium arsenate - Physico-chemical Properties
| Molecular Formula | AsH2NaO4 |
| Molar Mass | 163.92485 |
| Appearance | Solid |
| Use | Used as a reagent for the manufacture of other arsenic salts |
Monosodium arsenate - Risk and Safety
| UN IDs | 1685 |
| Hazard Class | 6.1(a) |
| Packing Group | II |
Monosodium arsenate - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Use | as a reagent for the manufacture of other arsenic salts |
| production method | production method a 5/3mol/L aqueous solution of arsenic acid is neutralized with a 5% aqueous solution of sodium hydroxide until the methyl orange indicates an equivalent point. The solution was evaporated by heating again until crystals formed. The cooled crude crystals were dissolved in water and saturated at 100 ° C. (solubility of 75.3g in g of water). The saturated solution was cooled to 0 °c with stirring. The precipitated colorless crystalline precipitate was suction-filtered, washed with ice water, and dried in a phosphorus pentoxide dryer. A monohydrate may be prepared. The water of crystallization is lost by heating to 100-130 °c. |
| category | toxic substances |
| toxicity grade | highly toxic |
| Acute toxicity | intravenous-rabbit LDL0: 45 mg/kg |
| flammability hazard characteristics | non-combustible; Thermal decomposition of toxic arsenide and sodium oxide smoke |
| storage and transportation characteristics | warehouse ventilation and low-temperature drying; Separate from food storage and transportation |
| fire extinguishing agent | water mist, sand |
| Occupational Standard | TWA 0.5 mg (arsenic)/m3; Tel 0.6 mg (arsenic)/M3 |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: Sodium arsenate monobasic Request for quotation
CAS: 10103-60-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 10103-60-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Sodium arsenate monobasic Request for quotation
CAS: 10103-60-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 10103-60-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History
